
UBT’s MammoWave® addresses the need for better breast cancer screening techniques, providing a safer and cost-effective alternative as a complementary imaging modality to mammograms. Copper Mountain Technologies’ 2-port C1209 9 GHz VNA is at the core of the MammoWave® system. The data from C1209 measurements, mainly complex S21 measurement, is processed through UBT’s dedicated software, helping to detect cancer cells. The initial version of the system is intended to be a quick, easy to use, and repeatable screening device. “Our mission is to allow ALL women to get screened,” explains Gianluigi Tiberi, UBT’s Head of R&D and co-founder.
Umbria Bioengineering Technologies (UBT), born as an academic spin off from the Department of Physics and Geology of the University of Perugia, Italy, is a biomedical company which develops a portfolio of innovative medical imaging devices based on cutting-edge microwave technology instead of dangerous ionizing radiation (X-Rays). The innovative imaging technology employs low power microwaves. To date, UBT developed two different products:
MammoWave, a novel X-Ray free mammogram for breast cancer screening.
BrainWave, a novel device for brain stroke detection and classification.
Currently, the most commonly used method of breast cancer detection is X-ray mammography. A mammogram is an X-ray examination of the breast and although it has been a fairly effective screening test, it has some drawbacks. X-rays are generally safe, but still carry a risk, especially with excessive exposure. Discomfort and pain are often associated with mammograms and there are limits to their effectiveness, particularly for younger women. UBT’s MammoWave® is a microwave mammogram for breast cancer detection, an X-ray free, comfortable, and effective alternative to X-ray mammography.
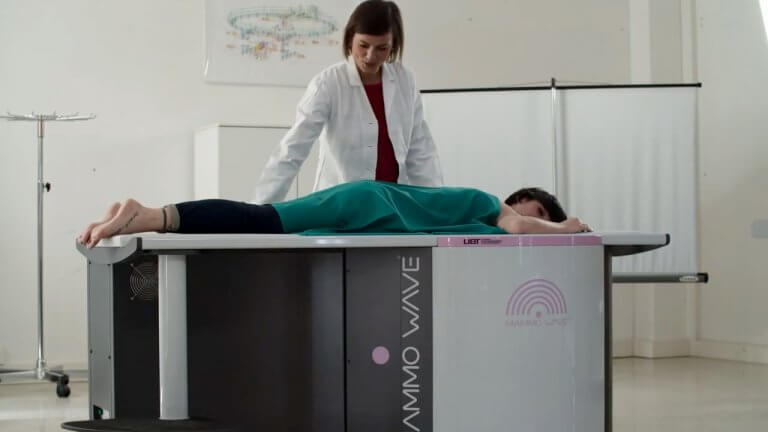
The microwave measurements for MammoWave® are driven by Copper Mountain Technologies’ Cobalt C1209 high-performance VNA, determining the difference between a healthy area and a possible sick zone. MammoWave® utilizes two antennas and each is connected to a port of the VNA. Screening is performed with the patient laying in a face down position. The breast is placed within a cup. The antennas rotate around the cup, without any compression, performing measurements approximately every four degrees. The measurements from the VNA are then processed through UBT’s software to detect the presence of suspected tumors in the breast tissue. Future developments of this technology may also be capable of drawing a 3D image to localize the area where cancer is possibly present.

UBT was in search of a VNA that could be easily integrated with their device. They needed a USB controlled instrument with fast measurement speed and high dynamic range. They were referred to the Cobalt C1209 9 GHz VNA by a representative at TEST Srl, Copper Mountain Technologies’ exclusive channel partner in Italy. When asked what aspects of the C1209 appealed to UBT’s team, CTO and co-founder Giovanni Raspa pointed to the performance and convenience offered by CMT: “The technical capability and the fact that we could have a demo to make the first test and setup of our device were deciding factors.” The advanced specifications of the C1209, and support from CMT engineers, enabled UBT to achieve their objective, “Continuous support from both the distributor and the engineers at Copper Mountain Technologies have enabled the proper setup and integration of this device into MammoWave® technology,” says Raspa.
Horizon 2020, an EU research and innovation program, awarded UBT an SME Instrument Phase 2 grant for a two-year project beginning in October 2018 . During the SME Instrument Phase 2 project, MammoWave® is undergoing an international clinical study throughout Europe, including trials at prestigious hospitals in Italy, Spain, and Germany. The trials are ongoing, with sales of this new device projected to start by the end of 2020. The excellent sensitivity, the simple method that minimizesthe discomfort synonymous with mammograms, and the safety of the microwave imaging technique have MammoWave® poised for mass breast cancer screening
Applications
Follow the link to learn more about the solutions CMT offers for various applications, as well as an opportunity to tell us about your unique VNA applications.
VNA Suggestions
Fill out this form and tell us about your VNA needs so that we may continue to design forward-thinking instruments that expand your test and measurement capabilities.


